Psi analysis
Psi analysis
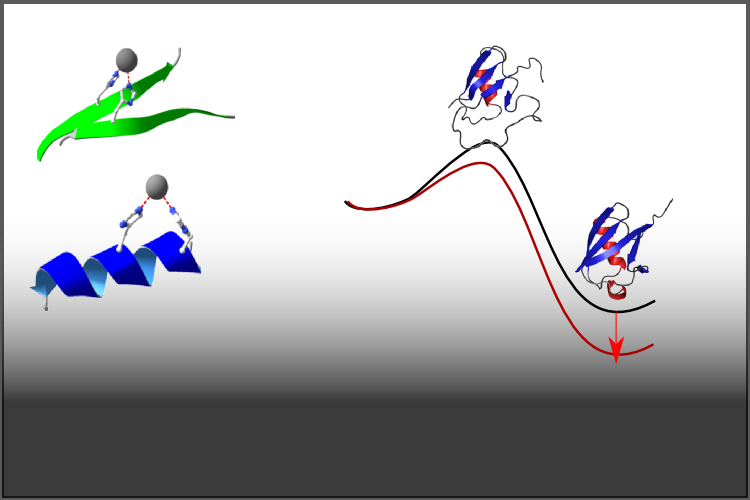
For many small single domain proteins, folding proceeds without the accumulation of any intermediates. It is possible though to probe the transition state by making perturbations and measuring the effects on the folding and unfolding rates.
A metal binding site can be introduced through mutation with 2 Histidine residues that in the native conformation is capable of binding a divalent metal ion, e.g. Zinc.
Measuring the effect of zinc on the folding and unfolding rates thus reports on the extent to which the two residues interact in the transition state.
Approach
- The folding transition states characterized using psi analysis are extensive with a highly native-like topology.